Discuss the article at www.lipid.org/lipidspin
Both genetic and observational studies have shown that elevated triglycerides (TG) are a marker of increased cardiovascular (CV) risk, even in statin-treated patients. (1) However, randomized clinical trials with TG-lowering drugs such as fibrates, niacin and low-dose omega-3 fatty acids (OM3FA) have failed to show any clinical benefit in patients already on statin therapy. Although these studies did not specifically target patients with elevated TG, post-hoc analyses in the subset of patients with elevated TGs suggest that lowering TG may improve cardiovascular outcomes.(2) The Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial (REDUCE-IT) was the first large randomized clinical trial to target high-risk patients on statin therapy who achieved low levels of low-density lipoprotein cholesterol (LDL-C) but still had elevated TG.(3) We review the findings of REDUCE-IT and discuss its implications in clinical practice.
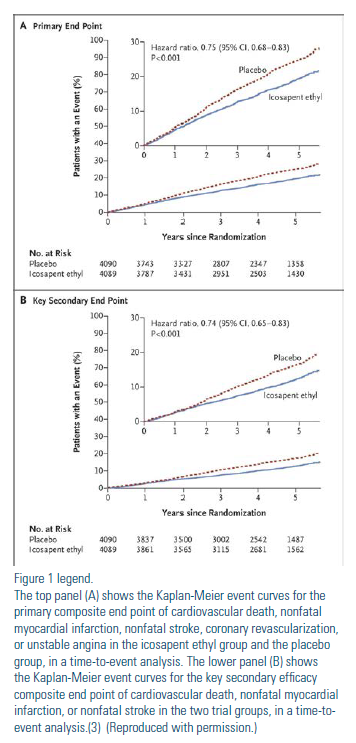
The results from REDUCE-IT are dramatic, especially when viewed in the context that the 25% reduction in major CV events was on a background of moderate- to high-intensity statin therapy and low levels of LDL-C. So how should a clinician integrate the findings from REDUCE-IT into clinical practice? For patients with elevated TG, lifestyle changes remain the first line of treatment. For high-risk patients – such as those with CV disease or diabetes who have persistent elevation in their fasting TG level, even if just borderline high (150 to 199 mg/dL) – highdose IPE (2 g twice daily) should be added to their statin therapy.
Although REDUCE-IT has answered an important question about the benefit of targeting elevated TG in high-risk statintreated patients, other questions remain. First, what is the lower threshold of TG levels in which high-dose IPE is of benefit? Although patients in REDUCE-IT could be enrolled with TG levels as low as 135 mg/dL, the median TG level in the study was much higher. Whether patients with TG levels below 150 mg/dL should be targeted with IPE therapy is unclear. The American Heart Association (AHA) defined an optimal TG level as < 100 mg/dL (4); whether this becomes our new target of therapy is yet to be determined. Second, the TG lowering in REDUCE-IT was modest and the 25% reduction in major CV events in the IPE group was greater than what would have been expected with this degree of TG lowering. Furthermore, the benefits of IPE were independent of the baseline or achieved TG levels. This suggests that other factors, such as IPE’s antiinflammatory and membrane stabilizing effects, may explain the reduction in CV events seen in REDUCE-IT. Will formulations of other prescription OM3FA that contain DHA but less EPA have the same beneficial effects as IPE? The Statin Residual Risk Reduction with Epanova in High Cardiovascular Risk Patients with Hypertriglyceridemia (STRENGTH) trial will help answer this question.(5) Third, will IPE show benefit in lower-risk primary prevention patients? Although the Vitamin D and Omega-3 Trial (VITAL), which used a low-dose OM3FA containing both EPA and DHA, was negative for its composite primary endpoint of major CV events, the risk of myocardial infarction was lower in the OM3FA compared to the placebo group.(6) Would high-dose IPE in a VITAL patient population have led to a positive outcome?
The landscape for lipid-lowering therapy is changing, and ongoing trials will help answer these questions. REDUCE-IT should quell any controversy about the benefits of targeting elevated TG in highrisk patients, even if the levels are just borderline high. In patients with CV disease or diabetes with elevated TG, 2 g of IPE twice daily should become standard add-on therapy to a maximally tolerated statin. The results from REDUCE-IT represent a sea change in secondary prevention and primary prevention in high-risk diabetic patients. REDUCE-IT has helped calm the waters, and we see nothing but smooth sailing ahead.
Disclosure statement: Dr. Slipczuk has no financial disclosures to report. Dr. Simlote has no financial disclosures to report. Dr. Karalis heceived honoraria from Sanofi/Regeneron.






.jpg)
.png)













