John is a 55-year-old man who heard on the news that all patients over 50 years old should be taking a statin. He is generally healthy, without high cholesterol, but with a C-reactive protein (CRP) >2. His primary care physician told him that he should watch his diet and exercise more. He would like a second opinion regarding the potential benefits of statins on his risk of having a heart attack or dying prematurely. Would John reduce his risk of major cardiovascular events and death by taking a statin rather than solely relying on diet and exercise? After conducting a literature search, you determine that the study most applicable to answering your patient’s therapy question is the Justification for the Use of Statins in Primary Prevention (JUPITER) trial,1 which compares rosuvastatin to placebo, in addition to diet/exercise. You recall that randomized controlled trials (RCTs) or systematic reviews are best for answering therapy questions. You recall that JUPITER is a double-blind, randomized trial with blinded outcomes assessors and <1 percent of patients lost to follow-up, but it ended early with only 1.8 years follow-up. Overall, its study design contains most elements that reduce the risk of serious bias. (Table 1) You decide to look at the results. JUPITER shows a 44 percent statistically significant relative reduction in risk of cardiac events with rosuvastatin and a 20-percent reduction in death, which seems pretty good. But you know this is a low-risk group given no history of heart disease and without elevated cholesterol, and you wonder how much absolute benefit John would actually have. You call on a friend, who directs you to the website ktclearinghouse.ca/cebm/toolbox/statscalc, which will easily calculate absolute and relative differences for you. Then you realize it is quite easy to calculate absolute differences yourself, since it is just the difference between the risk of events between the two groups. You find that the absolute reduction in cardiac events with rosuvastatin is 1.2 percent (2.8 to 1.6 percent) and the absolute reduction in death is only 0.6 percent (2.8 to 2.2 percent). Even though the relative reduction in risk is a good magnitude, you are less impressed by the absolute reductions, which seem pretty small. Plus, there are the potential harms to consider, particularly the development of new-onset diabetes, which has resulted in FDA-mandated monograph changes for statins. You see that the risk of new-onset diabetes in all patients is 0.6 percent higher with rosuvastatin.
You start thinking about another patient of yours, Max, who is higher risk because he already had a myocardial infarction. You definitely treat higher-risk patients like Max with statins, so you decide to review the supportive evidence you use in that treatment decision. You select the Scandinavian Simvastatin Survival Study (4S) trial, the classic landmark study of simvastatin in patients with existing heart disease. Having read the trial before, you recall that it is a double-blind, randomized trial with blinded outcomes assessors, no patients lost to follow-up, and five years of follow-up, a well-executed trial with low risk of bias. 4S shows a 30 percent statistically significant relative reduction in risk of cardiac events with simvastatin (lower than JUPITER), and a 29 percent reduction in risk of death (better than JUPITER). You calculate the absolute reduction in cardiac events in 4S of 6.7 percent and in death of 3.3 percent. These absolute numbers seem more impressive than those seen in a JUPITER populationtype patient. You note that few adverse effects were reported in 4S but that creatine kinase (CK) elevation >10x upper limit of normal occurred 0.23 percent more with simvastatin.

You start thinking about another patient of yours, Max, who is higher risk because he already had a myocardial infarction. You definitely treat higher-risk patients like Max with statins, so you decide to review the supportive evidence you use in that treatment decision. You select the Scandinavian Simvastatin Survival Study (4S) trial, the classic landmark study of simvastatin in patients with existing heart disease. Having read the trial before, you recall that it is a double-blind, randomized trial with blinded outcomes assessors, no patients lost to follow-up, and five years of follow-up, a well-executed trial with low risk of bias. 4S shows a 30 percent statistically significant relative reduction in risk of cardiac events with simvastatin (lower than JUPITER), and a 29 percent reduction in risk of death (better than JUPITER). You calculate the absolute reduction in cardiac events in 4S of 6.7 percent and in death of 3.3 percent. These absolute numbers seem more impressive than those seen in a JUPITER populationtype patient. You note that few adverse effects were reported in 4S but that creatine kinase (CK) elevation >10x upper limit of normal occurred 0.23 percent more with simvastatin.
You notice that the evidence-based medicine (EBM) website includes the calculation of number needed to treat (NNT) and number needed to harm (NNH), terms you have heard of but have not calculated yourself. You understand that number needed to treat is the number of patients needed to be treated under similar conditions as in the respective clinical trial to avoid one negative event. It seems relatively easy to calculate, simply taking the inverse of the absolute reduction in risk. So, for the 4S trial results, you find that for every 15 patients treated with simvastatin instead of placebo, one cardiac event will be prevented (1/0.067), and for every 30 patients treated with simvastatin instead of placebo, one death will be prevented (1/0.033). The number needed to harm for myopathy from the 4S study is 444 patients (Table 2). So it appears that only a small number of high-risk patients need to be treated before one benefits, while many are exposed before serious myopathy effects may occur. Numerically, the balance clearly favors the potential benefits over the risks in the 4S trial. Although, of course, you need to discuss with your patient and get his or her commitment to the therapy or run the risk of medication non-adherence, which we know is high with statins.
Now, turning back to JUPITER and your patient, John, who asked you whether a statin would decrease his risk of having a heart attack or dying prematurely more than diet and exercise would. You calculate an NNT of 84 for cardiac events and 167 for death, and an NNH of 167 for risk of new-onset diabetes. As you closely examine the numbers you calculated in Table 2, you notice that you would need to treat many more low-risk patients like John with a statin to prevent a cardiac event or death than high-risk patients like Max. While you intuitively knew this already, putting explicit numbers to the patient cases makes the difference in absolute size of effect more obvious to you. You realize that the relative reductions in risk that looked so favorable for both studies were misleading when considered in isolation. Since the benefit-risk balance is not as obvious as in a high-risk patient, you want to engage in a higher level of shared decision-making with John incorporating his values and preferences.
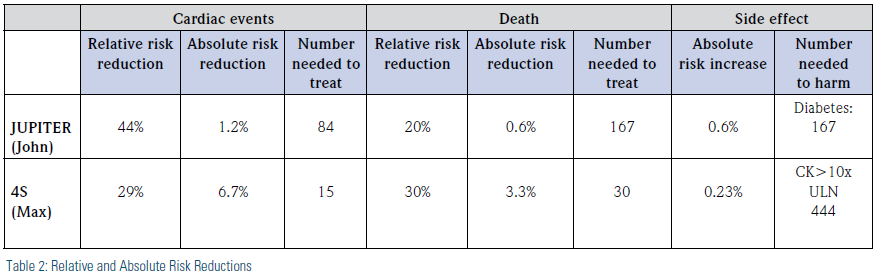
Overall, the numbers still seem to favor offering treatment to a low-risk patient like John, however, you decide that you will need to let him know the balance between the benefits and the harms of statin therapy. You let John know that, based on well-designed clinical trials, if someone already had heart disease, then only 15 patients would need to be treated before a serious cardiac event was prevented. However, in his case, since he is at low risk for heart disease, 84 patients like him would need to be treated with a statin before one patient would avoid having a serious cardiac event such as myocardial infarction, and the chance of having this benefit is twice as high as having a harm, such as developing diabetes. John says he would really prefer to try more intensive diet and exercise first because of the potential risk of developing new-onset diabetes, and he thanks you for discussing the details with him.
You reflect back on your work and realize that you have learned a lot about how to express the magnitude of the study findings, and have found that calculating both absolute and relative reductions in risk presents a more complete picture that has been helpful in considering the treatment decision questions for your patient. On reflection, you decide that you know some information about what biases clinical trials but realize you need a refresher on study design. You decide to do some further reading online at the JAMA website of the EBM Users’ Guide at jamaevidence.com.
These examples illustrate the practice of EBM, which includes the following steps:
- create an answerable clinical question
- search for the best evidence
- critically appraise the evidence
- study design (assessing bias in the methods)
- results (size of results and precision of results)
- apply the evidence to your patient
- self-assess your skills and abilities
In essence, the goal of EBM is to translate the best evidence into practice. In considering the treatment decisions for these patients, you have incorporated the three key aspects of EBM: the best evidence, your patient’s values and preferences, and your clinical expertise. While you have been somewhat reluctant to use the term “evidencebased medicine,” since it has been used and misused to the degree that almost everything is called evidence-based medicine or EBM these days, you like the idea of having this explicit framework to help you with your clinical decisionmaking. You found that there are many tools available for each of the steps of EBM and that you have just scratched the surface on how to express study results in a more meaningful way. Once you get the hang of these simple calculations, you are excited to move on to further EBM tools to help facilitate your patient care.
Disclosure statement: Dr. Jackevicius has received honorarium from the ACC and a grant from the AHA. Dr. Gupta has received consulting fees from AstraZeneca.
References are listed on page 35 of the PDF.






.jpg)
.png)












