Evidence in support of statins for the primary prevention of atherosclerotic cardiovascular disease (ASCVD) in women was sparse until recently.1 However, meta-analyses have documented that statins significantly reduce ASCVD event rates in women with elevated baseline risk.2,3,4
Yet a large subset of women under age 70 with hypercholesterolemia but no other risk factors does not reach the current threshold for statin eligibility, i.e., 7.5% absolute ASCVD risk in 10 years, even when total cholesterol elevation is severe.5 The reason is that 10-year risk prediction as currently recommended by the American College of Cardiology/American Heart Association (ACC/AHA) for primary prevention patients ages 40 to 75 years without diabetes or LDL-C > 190 mg/dl,6 and optionally by the National Lipid Association for those with 2 risk factors,7 is weighted heavily by age and less so by the absolute level of LDL-C. Thus, older women (and men) with relatively low LDL-C usually will be statin-eligible because 10-year ASCVD risk almost always exceeds 7.5%, while those under age 65 with LDL-C levels up to 189 mg/dl but 10-year risk under 7.5% will be ineligible. The fact that almost one-third of ASCVD events in women (and one-half in men) occur before the age of 65 highlights the fundamental limitation of this approach.8
As the optimal timing of statin initiation for primary prevention is being debated, a new strategy that accounts for the expected individual benefit from statin therapy based on clinical trial data has emerged that would expand treatment to a significant number of non-elderly, primary prevention women with hypercholesterolemia.
The individual-benefit model for statin allocation first was illustrated by Soran, et al., in 2014.9,10 The authors showed mathematically that the number of events prevented by statin therapy is a function of both 10-year ASCVD risk and baseline LDL-C. As such, those with a higher baseline LDL-C but lower 10-year risk can obtain an equivalent or greater benefit from statin therapy, i.e., a higher absolute risk reduction (ARR) and a lower number needed to treat (NNT), as those with a higher 10-year risk but lower LDL-C. That is because the magnitude of LDL-C lowering is a function of both statin potency and baseline LDL-C. At a given dose of a statin that lowers LDL-C by 43% (e.g., atorvastatin 20 mg), those with a higher LDL-C will experience a greater magnitude of LDL-C lowering than those with a lower baseline LDL-C. This will translate into a greater relative risk reduction (RRR) because the hazard ratio (HR) based on Cholesterol Treatment Trialist Collaboration meta-analytic data is 0.78 for every 1 mmol (39 mg/dl) of LDL-C lowering that occurs.11
Therefore, those with a higher baseline LDL-C who achieve a 2 mmol or 78 mg/dl reduction in LDL-C will have an HR of 0.78 x 0.78, or 0.60, or a 40% relative RR, while those with an even higher LDL-C who achieve a 3 mmol or 117 mg/dl reduction in LDL-C will have an HR of 0.783 or 0.47, or a 53% relative RR. This then can be multiplied by absolute risk in 10 years to calculate the number of events prevented per 100 patients over 10 years, as follows: # of events prevented/100 = [1 - 0.78n] x absolute 10-year risk, where n = LDL-C decrease in mmol/L.
For example, if ASCVD risk is 10% over 10 years and baseline LDL-C is relatively low and drops by 1 mmol/L or 39 mg/dl with statin therapy, the number of CVD events prevented per 100 people treated for 10 years is (1 - 0.783) × 10 = 2.2. ARR in 10 years as a percentage then is 2.2% or 0.022, and the NNT to prevent 1 event is 1/0.022, or 45. (Alternatively, the NNT = 100/2.2). On the other hand, if 10-year ASCVD risk is just 5% but LDL-C is high at baseline and lowered by 3 mmol/L, the number of events prevented per 100 people = (1 - 0.783 ) x 5, or 2.6, and the ARR over 10 years will be 2.6% and NNT 38, an even more cost-effective undertaking.
As seen in Table 1, it then follows that, at a given level of absolute risk, NNT by pre-treatment LDL-C, and guideline algorithms should consider adoption of the NNT in statin decision-making. Moreover, abolishing LDL-C therapeutic goals puts at a disadvantage those people with higher LDL-C levels who derive greater benefit from treatment aimed at specific targets.5
In 2016, Thanassoulis, et al., modeled statin eligibility based on individual benefit vs. risk-based strategies using a nationally representative sample of 2,134 primary prevention individuals age 40-75 from the National Health and Nutrition Examination Survey (NHANES) for the years 2005 to 2010.12
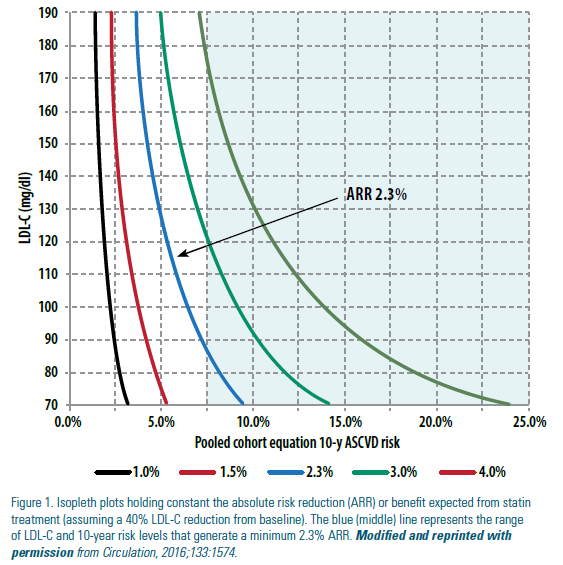
An individualized ARR of >2.3% over 10 years (corresponding to a NNT of 44 or less) was used as an acceptable cut-off for statin eligibility. The authors plotted the 10-year predicted risk and baseline LDL-C levels required to produce a constant individual benefit at pre-specified ARR levels (See Figure 1). As shown, a minimum ARR of 2.3% in 10 years (blue or middle curve in Figure 1) was met across a range of baseline LDL-C and 10-year risk levels, from an LDL-C of 189 mg/dl with a 10-year risk of <5% to an LDL-C of 70 mg/dl with a 10-year risk of just under 10%. On this basis the authors showed that the benefit model applied to their NHANES sample would extend statin treatment to 24.6 million Americans, 9.5 million more than the current ACC/AHA risk-based approach, and 60% of those newly statin-eligible would meet criteria for at least 1 statin trial (Figure 2). For the entire 24.6 million identified via the benefit-based model, the average 10-year ASCVD risk, 10-year ARR, and NNT were 10.9%, 4% and 25 (range 9-44), respectively, compared to 13.9%, 4.8% and 21 (range 9-44), respectively, for the 15 million targeted via the risk-based approach. As expected, the 9.5 million additional statin-eligible Americans were significantly younger (mean age 55.2 versus 62.5 years) and had a significantly higher baseline LDL-C (140 vs. 133 mg/dL). Moreover, a significant proportion (about one-third) were women, and their proportional increase in statin eligibility was at least as high as for men.13
The authors further calculated that benefit- based statin eligibility would be expected to prevent 999,080 ASCVD events over 10 years, or 266,508 more than the risk-based model.
In summary, 10-year risk-based strategies limit statin eligibility in primary prevention women under age 65 with elevated LDL-C but levels less than 190 mg/dl. A benefit-oriented approach that bases statin treatment on the expected ARR from LDL-C lowering would extend statin treatment to a significant number of younger hypercholesterolemic primary prevention women with absolute 10-year risk under 7.5% but elevated future risk. As such it would help “level the playing field” and refocus treatment on the root cause of ASCVD. As noted by Thanassoulis, et al., this strategy also may be better aligned with the new paradigm of personalized or precision-based cardiovascular medicine,14 and with attempts to “harmonize” ASCVD risk reduction interventions.15
Disclosure statement: Dr. Aspry has conducted industry sponsored research with Amgen and Akcea.
References available here.






.jpg)
.png)











.PNG)